The Atom
What is an atom?
What are atoms made of?
* Atoms are the basic building blocks of ordinary matter.
* Atoms can join together to form molecules, which in turn form most of the objects around you.
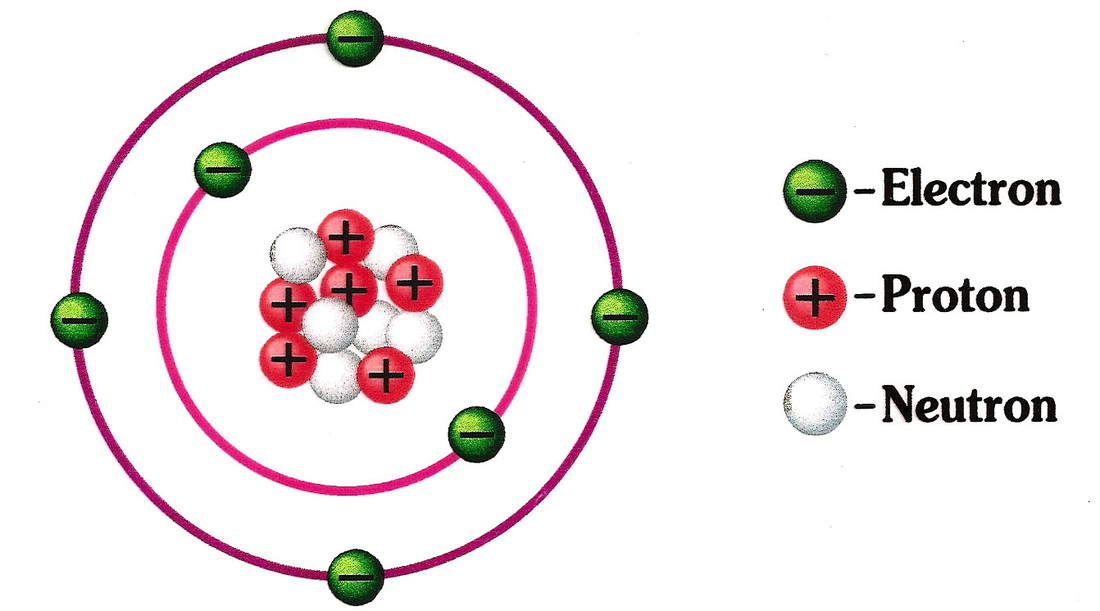
* Atoms are composed of particles called protons, electrons and neutrons.
* Protons carry a positive electrical charge, electrons carry a negative electrical charge and neutrons carry no electrical charge at all.
The protons and neutrons cluster together in the central part of the atom, called the nucleus, and the electrons 'orbit' the nucleus.
A particular atom will have the same number of protons and electrons and most atoms have at least as many neutrons as protons.
The Periodic Table
The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number (number of protons), electron configurations, and recurring chemical properties.
This ordering shows periodic trends, such as elements with similar behavior in the same column. It also shows four rectangular blocks with some approximately similar chemical properties.
In general, within one row (period) the elements are metals on the lefthand side, and non-metals on the righthand side.
The rows of the table are called periods; the columns are called groups.
The periodic table can be used to derive relationships between the properties of the elements, and predict the properties of new elements yet to be discovered or synthesized.
Dmitri Mendeleev published in 1869 the first widely recognized periodic table. He developed his table to illustrate periodic trends in the properties of the then-known elements. Mendeleev also predicted some properties of then-unknown elements that would be expected to fill gaps in this table. Most of his predictions were proved correct when the elements in question were subsequently discovered. Mendeleev's periodic table has since been expanded and refined with the discovery or synthesis of further new elements and the development of new theoretical models to explain chemical behavior.
All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been discovered or synthesized, with elements 113, 115, 117, and 118 being confirmed by the IUPAC on December 30, 2015.[1]
The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature. Elements with atomic numbers from 95 to 118 have only been synthesized in laboratories. It has been shown that elements 95 to 100 once occurred in nature but currently do not.
Synthesis of elements having higher atomic numbers is being pursued. Numerous synthetic radionuclides of naturally occurring elements have also been produced in laboratories.
The Periodic Table
Video
"Meet the Elements"
Video
Metals and Non-metals
Video
Guide Questions
| chapter_12__guide_questions_for_weebly.pdf | |
| File Size: | 133 kb |
| File Type: | |